Photo Credit: Centers for Disease Control and Prevention's DPDx Parasitology Diagnostic Web Site http://www.dpd.cdc.gov/dpdx/HTML/ImageLibrary/Malaria_il.htm
Plasmodium falciparum[]
Plasmodium falciparum is an Apicomplexan (phylum that includes organisms with the organelle called an apicoplast and an apical complex). It is responsible for 95% of the deaths caused by malaria (Dr. Joseph Schall's Biology 246 lecture, 2013) and is the most lethal of all the human malaria parasites.
Plasmodium falciparum is found to be unique compared to the other malaria parasites because it undergoes sequestration (it is able to adhere to the endothelial cells of the capillaries). It is also distinctive due to the banana or crescent shape that its gametocytes have (Dixon et al., 2012). Plasmodium falciparum is known to cause malignant tertian malaria w hich means that every 42-47 hours you go through a cycle of fevers (Allison Neal's Biology 246 Lab lecture).
Life Cycle[]
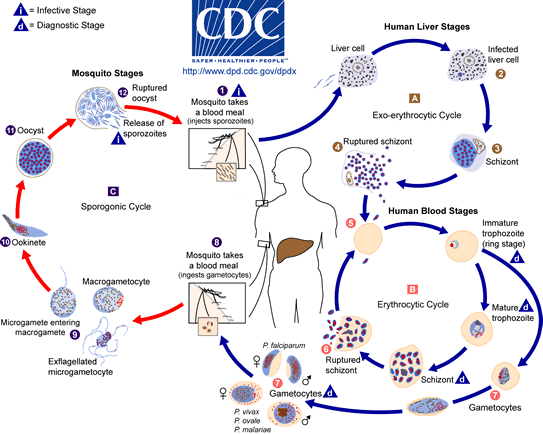
Sporozoites (infective stage) are injected into a human host from the salivary glands of a mosquito (of the genus Anopheles) when the mosquito takes a blood meal. The sporozoites penetrate liver cells and go through a cycle of schizogany where the sporozoites develop into schizonts (asexual replication stage). The liver cells eventually burst to release the many merozoites that have developed in the schizont. The merozoites penetrate red blood cells. There, the merozoite becomes a ring-form trophozoite (feeding stage named for its ring-like shape). In the red blood cells, the parasite goes through another cycle of schizogany. A schizont develops and eventually the red blood cell bursts to release many trophozoites. Some of these trophozoites will go through erythrocytic schizogany again while a few will develop into male and female gametocytes in new red blood cells. At this point, the gametocytes can be picked up by another mosquito who takes a blood meal. In the mosquito, the male gametocyte develops flagella and then exflagellates. Each flagella can fertilize a female gametocyte. The fertilized stage is called an ookinete. The ookinete finds its way to the midgut of the mosquito where it attaches and develops into an oocyst (stage containing sporozoites). The oocyst eventually bursts and the sporozoites travel to the salivary glands of the mosquito. Now, the parasite is ready to be transmitted to another human when the mosquito takes another blood meal (Dr. Joseph Schall's Biology 246 lecture).
See a video representation of the malaria life cycle at http://www.proteinlounge.com/Animation/Malaria.
Recent Research[]
An interesting study published in 2013 examines the antimalarial activity of certain glycosphingolipids (constituents of cell membranes) isolated from an African marine sponge, Axinyssa dijiferi, that inhabits mangrove tree roots. The glycosphingolipids' (named axidijiferosides) antimalarial effects were tested on a chloroquine-resistant strain of Plasmodium falciparum. The IC50 (concentration of a drug needed to inhibit 50% of a biological process) of the axidijiferosides were 5x less than chloroquine's but they showed low cytotoxicity and were specific to malaria (no activity shown against other parasites such as Leishmania donovani or Trypanosoma brucei) (Farokhi et al., 2013).
Another study done by Tiburcio et al. shows that as Plasmodium falciparum gametocytes mature, less STEVOR proteins are expressed which correlates to increased deformability of the erythrocytes they infect. The authors suggest that the deformability of erythrocytes containing mature gametocytes allows these infected erythrocytes to escape retention by the spleen by maneuvering through sinusoids (capillaries that are discontinuous aka have openings). It is also suggested that these proteins may be a possible future target for decreasing transmission of malaria by not allowing the mature gametocytes to escape into circulation where they can be taken up in a blood meal by a mosquito (Tiburcio et al., 2012).
Plasmodium falciparum has increasingly become more resistant to the antimalarial drug, chloroquine, which works by disrupting the parasite's mechanism to detoxify the heme molecules resulting from the parasite's digestion of host hemoglobin. Researchers are working on discovering the specific mechanism that now allows P. falciparum to avoid the remedial effects of chloroquine. Fidock et al. used PCR amplification with primers to locate the genetic sequences in the cDNA library of the P. falciparum genome. They found mutations in the pfcrt gene that codes for a new transmembrane protein which seems to confer chloroquine resistance to the individuals that have it (Fidock et al., 2000).
References[]
Dixon M.W.A., et al. 2012. Shape-shifting gametocytes: how and why does P. falciparum go banana-shaped? Trends in Parasitology 28(11): 471-478.
Farokhi F., et al. 2013. Antimalarial Activity of Axidijiferosides, New β-Galactosylceramides from the African Sponge Axinyssa dijiferi. Marine Drugs 11: 1304-1315.
Fidock D.A., et al. 2000. Mutations in the P. falciparum Digestive Vacuole Transmembrane Protein PfCRT and Evidence for Their Role in Chloroquine Resistance. Molecular Cell 6: 861-871.
Tiburcio, M., et al. 2012. A switch in infected erythrocyte deformability at the maturation and blood circulation of Plasmodium falciparum transmission stages. Blood 119 (24): 172-180.


